Scroll to:
Self-propagating high-temperature synthesis of a highly dispersed Si3N4–SiC ceramic powders composition using sodium azide and polytetrafluoroethylene
https://doi.org/10.17073/1997-308X-2025-3-25-38
Abstract
Refractory ceramic composite materials of the silicon nitride–silicon carbide (Si3N4–SiC) system possess a wide range of valuable properties and are used across various industrial fields as excellent refractories, structural heat-resistant materials capable of withstanding high mechanical loads at elevated temperatures, and lightweight functional materials for microwave radiation shielding in aviation and aerospace applications. The performance of Si3N4–SiC composite ceramics can be significantly enhanced by increasing the dispersion of the component powders, transitioning from micron-sized particles to highly dispersed powders (<1 µm). This study focuses on improving a simple, energy-efficient method of azide self-propagating high-temperature synthesis (SHS) for obtaining such highly dispersed powder compositions, using mixtures of sodium azide (NaN3) with elemental silicon and carbon powders, activated and modified by carbiding addition of powdered polytetrafluoroethylene (PTFE). These charge compositions, in both bulk and pressed forms, were combusted in a nitrogen atmosphere at 3 MPa. The maximum pressure and solid product yield were measured. The phase composition and microstructure of the combustion products were examined using X-ray diffraction (XRD) and scanning electron microscopy (SEM). The introduction of PTFE as a reactive carbiding and activating additive effectively overcame the limitations of conventional azide SHS processes that use halide salts such as NH4F, Na2SiF6 , and (NH4)2SiF6 . In addition to ensuring a high dispersion of the synthesized powders, the phase composition – particularly for the pressed charges – became significantly closer to the target theoretical composition. Notably, the silicon carbide content in the Si3N4–SiC product increased substantially, while the amounts of free silicon and carbon impurities decreased.
Keywords
For citations:
Uvarova I.A., Amosov A.P., Titova Yu.V., Novikov V.A. Self-propagating high-temperature synthesis of a highly dispersed Si3N4–SiC ceramic powders composition using sodium azide and polytetrafluoroethylene. Powder Metallurgy аnd Functional Coatings (Izvestiya Vuzov. Poroshkovaya Metallurgiya i Funktsional'nye Pokrytiya). 2025;19(3):25-38. https://doi.org/10.17073/1997-308X-2025-3-25-38
Introduction
Refractory ceramic composite materials of the silicon nitride–silicon carbide (Si3N4–SiC) system possess a wide range of valuable properties and are used in various industrial applications. Primarily, these materials are excellent refractories and can be used at high temperatures (up to 1500 °C) due to their outstanding oxidation and creep resistance, chemical stability, low thermal expansion coefficient, and thermal shock resistance [1]. Such refractories are typically produced by reaction bonding of SiC powder with Si3N4 particles synthesized via direct nitridation of silicon in a pressed powder mixture of SiC and Si under a nitrogen atmosphere at elevated temperatures (1300–1450 °C) for several hours [1; 2]. The resulting product is a two-phase mixture of α-Si3N4 and β-Si3N4 . The density and mechanical strength of the composites increase with a higher content of the fibrous α-Si3N4 phase, which is promoted by reducing the particle sizes of SiC and Si, as well as by the presence of trace oxygen impurities in the nitrogen atmosphere. This oxygen leads to intensive oxidation of silicon, formation of SiO vapor, and gas-phase synthesis of α-Si3N4 [1; 2]. While commercially produced silicon carbide refractories (without Si3N4 in their composition) exhibit a compressive strength of up to 100 MPa at approximately 20 % porosity [3], a reaction-bonded composite with a composition of 46SiC–50Si3N4–4Y2O3 (wt. %) demonstrates a compressive strength of 319 MPa at 26 % porosity [1].
Si3N4–SiC composite refractories are also produced by other methods such as semi-dry pressing (with up to 8 % liquid binder), plastic forming (up to 24 % binder), and slip casting (with up to 80 % water), followed by drying and sintering at temperatures up to 1600 °C [4–6]. In these methods, the starting materials are typically mixtures of polydisperse Si3N4 and SiC micropowders, with particle sizes ranging from fine fractions (1–5 µm) to coarse ones (50–200 µm), to achieve optimal packing density in both the powder mixture and the final composite. However, the use of such relatively coarse powders, combined with a porosity of approximately 20 %, limits the ability of Si3N4–SiC refractories to reach their maximum mechanical strength, particularly at elevated temperatures.
To use Si3N4–SiC composites as structural heat-resistant materials under high mechanical loads at elevated temperatures (e.g., in gas turbine engines), or as ultra-hard tooling and wear-resistant materials, it is essential to achieve the highest possible values of hardness, strength, and fracture toughness. To achieve near-theoretical density and reduce porosity to below one percent, alternative fabrication methods must be employed. These include pressureless sintering of Si3N4 and SiC powder mixtures at temperatures up to 2000 °C (including liquid-phase sintering), hot pressing, hot isostatic pressing, spark plasma sintering, and additive manufacturing techniques [5; 7–10]. Numerous studies utilizing these methods have shown that the mechanical properties of Si3N4–SiC composite ceramics improve significantly with increased fineness of the constituent powders – that is, as particle sizes are reduced from the micron scale to highly dispersed (<1 µm), submicron (0.1–1.0 µm), and even nanoscale (1–100 nm) dimensions. Consequently, considerable research efforts are currently focused on the development of nanostructured Si3N4–SiC ceramic composites [11–15]. In addition to enhanced mechanical performance, Si3N4–SiC nanocomposites exhibit improved microwave absorption properties, making them promising candidates for use as lightweight functional materials for electromagnetic shielding in aerospace and aviation applications [16].
At first glance, the ex situ fabrication of Si3N4–SiC nanocomposites – based on mechanical mixing of pre-synthesized Si3N4 and SiC nanopowders followed by compaction and sintering – appears to be the most straightforward approach. However, such nanopowders are extremely costly, as they cannot be produced by simple mechanical milling of inexpensive coarse powders, but instead require synthesis via various complex and expensive chemical routes [5]. Furthermore, nanoparticles exhibit pronounced interparticle adhesion, forming dense agglomerates that are difficult to disperse through mechanical mixing, thereby impeding the formation of a homogeneous nanopowder blend. This lack of homogeneity often results in structural defects during densification and sintering, including chemical inhomogeneity, grain size non-uniformity, porosity, and crack formation [14]. From this standpoint, when dealing with highly dispersed powders, in situ chemical synthesis methods are preferable. These approaches enable the direct formation of the desired ceramic powder composition from precursor reagents, eliminating the drawbacks associated with ex situ mechanical mixing of pre-synthesized ceramic powders [1; 14].
Several chemical synthesis methods have been developed for producing submicron and nanopowder compositions of Si3N4–SiC. These include pyrolysis of silicon-containing polymers, methane pyrolysis followed by coating of Si3N4 particles with highly dispersed carbon, carbothermal reduction of silicon dioxide in the presence of gaseous nitrogen, gas-phase reactions, and plasma-chemical synthesis [14–21]. However, these methods are characterized by high energy consumption due to the need for elevated temperatures and prolonged heat treatment, as well as the use of expensive equipment and reagents. As a result, such techniques remain largely confined to laboratory-scale production of composite powders and have not yet been transferred to industrial-scale manufacturing. Currently, commercial suppliers of ceramic powders continue to produce primarily single-phase powders, and most nanocomposite ceramic powders are still obtained by conventional methods involving the mixing and milling of their constituent single-phase powders [14; 15]. Nevertheless, chemical synthesis methods for composite powders are regarded as advanced and promising, warranting further development and industrial implementation. Once this goal is achieved, high-quality nanocomposite powders will become commercially available, which is expected to have a positive impact on the performance characteristics of the resulting composite ceramics [14; 15].
Among the chemical methods used to synthesize submicron and nanoscale Si3N4–SiC powder compositions, self-propagating high-temperature synthesis (SHS) stands out for its simplicity and energy efficiency. This method is based on the combustion of inexpensive precursor reagents [22–24]. In the simplest case, combustion of a powder mixture of silicon (particle size d < 15 µm) and technical carbon (d < 1 µm) in gaseous nitrogen at a pressure of 1–7 MPa – that is, in the Si–nC–N2 system – can be initiated when the carbon black content does not exceed n = 0.8. The combustion product is a Si3N4–SiC powder composition containing 5–60 wt. % SiC, with the remainder comprising silicon nitride predominantly in the β-phase, along with a minor amount of unreacted silicon. Most of the resulting particles are 2–3 µm in size, though larger particles up to 15 µm are also present, typically in the form of columnar crystals or agglomerates of finer particles.
The addition of powdered polytetrafluoroethylene (PTFE) (C2F4)n in small amounts (5–15 wt. %) acts as an activating agent, extending the combustion limits of the Si–C–N2 system and enabling the synthesis of composites with a wide range of Si3N4 : SiC ratios (from 0 to 100 %). The resulting powders exhibit micron-scale particle sizes and a low fraction of the α-Si3N4 phase [22]. A more advanced approach involves azide self-propagating high-temperature synthesis (azide SHS), applied to systems based on Si–C–NaN3–halide salts. In this method, silicon (Si), technical carbon (C), sodium azide (NaN3 ) as the nitriding agent, and halide salts (NH4F, Na2SiF6 , and (NH4)2SiF6 ) as activating, gasifying additives are used. Combustion of these bulk powder mixtures in a 4.5 L reactor under 4 MPa of nitrogen produces highly dispersed Si3N4–SiC powder compositions containing 1.6–41.8 wt. % SiC. Compared to earlier combustion-based SHS products, these compositions feature finer particle sizes (mainly in the range of 100–500 nm) and a markedly higher content of the α-Si3N4 phase – up to ten times greater than that of β-Si3N4 [23–25]. However, the experimental compositions deviated significantly from the theoretical ones, with an excess of silicon nitride, a reduced fraction of silicon carbide, and notable levels of free silicon (up to 5.7 wt. %) and free carbon (up to 5.1 wt. %) as impurities.
It is well known that the synthesis reaction of silicon carbide from elemental silicon and technical carbon powders (Si + C = SiC) is weakly exothermic and cannot proceed in combustion mode under standard conditions [26]. However, the reactivity of the Si + C mixture can be enhanced through preheating, application of an electric field, mechanical activation, chemical activation, or by using a gaseous nitrogen or air environment, enabling the synthesis of SiC via combustion. One of the simplest and most effective approaches involves the use of polytetrafluoroethylene (C2F4)n as a chemical activator. This method enables complete reaction of the mixture Si + 0.9C + 0.05C2F4 → SiC + 0.1F2 in a nitrogen atmosphere at 3 MPa, resulting in the formation of SiC particles with an average size of ~200 nm [27]. The reaction stages and structure formation in the combustion wave of Si–C–C2F4 mixtures with relatively high PTFE content (C2F4 : C = 0.5÷3.0) were studied in [28] by burning pressed powder mixtures in an argon atmosphere at 0.5 MPa. The combustion products, in addition to silicon carbide, contained 16–33 % residual silicon (necessary for subsequent Si3N4 formation in Si3N4–SiC composite synthesis) and consisted of porous agglomerates 10–20 µm in size, incorporating crystallized silicon droplets and SiC grains measuring 0.3–0.7 µm. Furthermore, PTFE can be used not only as an activating additive but also as a carbiding reagent in place of technical carbon during the combustion of a bulk Si + C2F4 mixture in gaseous argon at 0.5 MPa and the synthesis of silicon carbide in the form of fibers (100–500 nm in diameter) and equiaxed particles (0.5–3.0 µm), which aggregate into porous agglomerates [29]. However, the overall SiC yield in this case is quite low – only about 10 % of the charge reacts to form cotton-like (fluffy) SiC, while the remainder consists of a black powder of unreacted carbon and silicon. This result is attributed to the following sequence of reactions involved in the formation of silicon carbide with PTFE (C2F4) [28; 29]:
| 2C2F4 → CF4(g) + 2CF2(g) + C(s), | (1) |
| 4Si(l) + 2CF4(g) + 2CF2(g) → SiF4(g) + 2SiF3(g) + SiF2(g) + 4C(s), | (2) |
| 2SiF2(g) + 2SiF3(g) → 2.5SiF4(g) + 1.5Si(l), | (3) |
| Si(l) + C(s) → SiC(s). | (4) |
At the first stage (1), PTFE undergoes exothermic decomposition in the preheating zone, producing gaseous fluorides and solid carbon particles. During the intermediate stages (2) and (3), the gaseous fluorides react with each other and with molten silicon. In the final stage (4), silicon and carbon particles – originating both from the initial soot and from PTFE decomposition – react to form the target silicon carbide. Completion of all these stages is essential to obtain highly dispersed SiC; if only the first stage occurs, the result is merely the formation of gaseous fluorides and soot particles [29]. Combustion involving PTFE proceeds at a high rate and is accompanied by intense gas evolution, resulting in the scattering of the charge components. Under these conditions, silicon particles are unlikely to react with carbon particles, as such a reaction is improbable in the gas phase [29]. Therefore, when using PTFE, the charge should be pressed into briquettes – rather than used in bulk form – preferably with a diameter of at least 30 mm, and the combustion should be conducted under elevated gas pressure in an SHS reactor to prevent the escape of PTFE decomposition products from the reaction zone [27–30].
As demonstrated in our previous study [31], this approach proved effective in the azide SHS of another highly dispersed powder composition, AlN–SiC, using PTFE. Partial replacement of carbon in the carbiding mixture with 0.9C + 0.05C2F4 eliminated, in most cases and across various target AlN/SiC phase ratios, the drawbacks associated with conventional azide SHS using halide salts such as (NH4)2SiF6 , AlF3 , and NH4F. While maintaining the high dispersity of the synthesized AlN–SiC powder compositions, their phase composition – especially when pressed charges were used – became significantly closer to the intended theoretical composition. The SiC phase content increased substantially, while undesirable secondary phases such as silicon nitride and the water-insoluble salt cryolite (Na3AlF6 ) were eliminated.
An attempt to synthesize the target stoichiometric composition of the highly dispersed Si3N4–SiC nitride–carbide system by fully replacing carbon with PTFE in the initial azide SHS reagent mixture was unsuccessful [32]. In bulk mixtures, only small amounts of the desired Si3N4 and SiC phases were formed, with free carbon being the predominant combustion product. This result aligns with the findings reported in [29] and is attributed to the combustion proceeding only through the initial stages – specifically, reactions (1) and (2), which involve PTFE decomposition and the generation of gaseous silicon fluorides and free carbon. In pressed mixtures, a substantial fraction of the silicon particles was able to react with carbon (reaction 4), leading to increased formation of the target Si3N4 and SiC phases. However, due to continued scattering of the charge components during combustion, a significant portion of the silicon remained unreacted. As a result, the amount of synthesized SiC was considerably lower than the theoretical value predicted by the stoichiometric equations.
In light of these findings and building upon previous studies [23–25; 32], the present work aimed to increase the SiC content and bring the composition of the highly dispersed Si3N4–SiC material closer to its theoretical target. To this end, we investigated the azide SHS process involving partial substitution of carbon with PTFE in the initial reagent mixture – specifically, the combustion process and resulting products of the Si–NaN3–C–C2F4 system.
Research methodology
To investigate the azide SHS process for synthesizing highly dispersed Si3N4–SiC compositions with partial substitution of carbon by PTFE in the initial charge (reagent mixture), the following starting materials were used (wt. % unless otherwise noted): silicon powder, grade Kr00 (≥99.9 %, d = 40 µm); sodium azide powder, analytical grade (≥98.71 %, d = 100 µm); polytetrafluoroethylene (PTFE), grade PN-40 (≥99.0 %, d = 40 µm); and technical carbon black, grade P701 (≥88.0 %, d = 70 nm, in the form of agglomerates up to 1 µm).
According to [22], achieving a high SiC content in silicon nitride-based composites requires partial replacement of technical carbon with an activating carbiding additive – polytetrafluoroethylene (PTFE) – in amounts of 5, 10, and 15 %. This yields carbiding mixtures of technical carbon and PTFE with the following compositions, each equivalent to 1 mol of carbiding carbon:
| 0.9C + 0.05C2F4 , | (A) |
| 0.8C + 0.1C2F4 , | (B) |
| 0.7C + 0.15C2F4 . | (C) |
In this azide SHS approach, the strongly nitriding reagent sodium azide (NaN3) is added to the charge in quantities sufficient to neutralize the fluorine released during the complete decomposition of PTFE and bind it as water-soluble sodium fluoride (NaF), which can subsequently be removed from the SHS product by washing with water. As a result, the stoichiometric equations for the azide SHS of Si3N4–SiC powder compositions for five molar ratios of the target phases (Si3N4 : SiC = 4:1, 2:1, 1:1, 1:2, 1:4) using carbiding mixtures (A)–(C) containing PTFE and combusted in a nitrogen atmosphere are as follows:
– for carbiding mixture (A):
| 13Si + 0.2NaN3 + 0.9C + 0.05C2F4 + 7.7N2 = 4Si3N4 + SiC + 0.2NaF, | (5) |
| 7Si + 0.2NaN3 + 0.9C + 0.05C2F4 + 3.7N2 = 2Si3N4 + SiC + 0.2NaF, | (6) |
| 4Si + 0.2NaN3 + 0.9C + 0.05C2F4 + 1.7N2 = Si3N4 + SiC + 0.2NaF, | (7) |
| 5Si + 0.4NaN3 + 1.8C + 0.1C2F4 + 1.4N2 = Si3N4 + 2SiC + 0.4NaF, | (8) |
| 7Si + 0.8NaN3 + 3.6C + 0.2C2F4 + 0.8N2 = Si3N4 + 4SiC + 0.8NaF; | (9) |
– for carbiding mixture (B):
| 13Si + 0.4NaN3 + 0.8C + 0.1C2F4 + 7,4N2 = 4Si3N4 + SiC + 0.4NaF, | (10) |
| 7Si + 0.4NaN3 + 0.8C + 0.1C2F4 + 3,4N2 = 2Si3N4 + SiC + 0.4NaF, | (11) |
| 4Si + 0.4NaN3 + 0.8C + 0.1C2F4 + 1,4N2 = Si3N4 + SiC + 0.4NaF, | (12) |
| 5Si + 0.8NaN3 + 1.6C + 0.2C2F4 = Si3N4 + 2SiC + 0.8NaF, | (13) |
| 7Si + 1.6NaN3 + 3.2C + 0.4C2F4 = Si3N4 + 4SiC + 1.6NaF + 0.4N2 ; | (14) |
– for carbiding mixture (C):
| 13Si + 0.6NaN3 + 0.7C + 0.15C2F4 + 7.1N2 = 4Si3N4 + SiC + 0.6NaF, | (15) |
| 7Si + 0.6NaN3 + 0.7C + 0.15C2F4 + 3.1N2 = 2Si3N4 + SiC + 0.6NaF, | (16) |
| 4Si + 0.6NaN3 + 0.7C + 0.15C2F4 + 1.1N2 = Si3N4 + SiC + 0.6NaF, | (17) |
| 5Si + 1.2NaN3 + 1.4C + 0.3C2F4 + 0.2N2 = Si3N4 + 2SiC + 1.2NaF, | (18) |
| 7Si + 2.4NaN3 + 2.8C + 0.6C2F4 = Si3N4 + 4SiC + 2.4NaF + 1.6N2 . | (19) |
The reagent mixtures corresponding to equations (5)–(19), each with an average mass of 22 g, were combusted in a 4.5 L azide SHS reactor under an initial nitrogen pressure of P0 = 3 MPa. Combustion was performed in two forms: as a bulk charge, placed in a tracing-paper crucible (30 mm in diameter and 45 mm in height), and as briquetted charges, compacted under 7 MPa into cylindrical pellets measuring 30 mm in diameter and approximately 22 mm in height. Combustion was initiated using a tungsten spiral heater. The maximum gas pressure (Pmax ), generated during combustion was recorded via a manometer. After cooling, the combustion products were removed from the reactor, ground into free-flowing powder using a porcelain mortar, and washed with water to remove the byproduct – sodium fluoride (NaF). The dried and washed combustion product was weighed, and the mass loss (∆m, %) was determined as the difference between the initial charge mass (m0 ) and the final product mass (mf ). This mass loss was interpreted as deviation of reactants and products caused by intense combustion. The phase composition of the synthesized products was analyzed using an ARL X’TRA powder X-ray diffractometer (Thermo Fisher Scientific, Switzerland) equipped with a copper anode X-ray tube. Diffraction patterns were processed and the quantitative phase composition determined by the Rietveld refinement method using HighScore Plus software and the COD-2024 crystallographic database. The morphology and particle size of the synthesized powders were examined using a JSM-6390A scanning electron microscope (JEOL, Japan).
Results and discussion
In chemical equations (5)–(19), the composition of the reaction products is expressed in moles, while in the experiments, it is given in weight percent. When converting the molar ratios of silicon nitride to silicon carbide to weight percentages, the following theoretical ratios of the target Si3N4–SiC compositions are obtained, assuming complete removal of the water-soluble byproduct sodium fluoride (NaF) from the products of reactions (5)–(19):
| 4:1 → 4Si3N4 + SiC = 93.3 % Si3N4 + 6.7 % SiC, | (20) |
| 2:1 → 2Si3N4 + SiC = 87.5 % Si3N4 + 12.5 % SiC, | (21) |
| 1:1 → Si3N4 + SiC = 77.8 % Si3N4 + 22.2 % SiC, | (22) |
| 1:2 → Si3N4 + 2SiC = 63.6 % Si3N4 + 36.4 % SiC, | (23) |
| 1:4 → Si3N4 + 4SiC = 46.7 % Si3N4 + 53.3 % SiC. | (24) |
The experimentally determined combustion parameters – including the maximum pressure in the reactor (Pmax ) and mass loss (Δm) – for the initial bulk and pressed powder charges corresponding to reactions (5)–(19), along with the phase compositions of the washed solid reaction products, are summarized in the Table.
Combustion parameters of the initial powder mixtures (charges)
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The Table shows that the bulk charges corresponding to reactions (5)–(8) did not combust, while combustion of the charge from equation (9) did not yield the target phases Si3N4 and SiC. Instead, the combustion products were merely a mixture of free silicon and carbon. These results can be explained by the fact that reactions (5)–(9) used carbiding mixture (A), which contains the lowest amount of the activating additive – polytetrafluoroethylene (0.05C2F4 per 1 mole of carbiding carbon). At the same time, these charges included a relatively large amount of poorly reactive silicon powder – ranging from 4 to 13 moles. Only the charge in equation (9), which had the highest PTFE content (0.2C2F4 ), was able to sustain combustion. However, under bulk conditions, this reaction yielded only a mixture of Si and C particles, consistent with the findings reported in [29]. This outcome is attributed to the progression of only reaction (1) – the exothermic decomposition of PTFE – as the first stage of the process. The table also shows that the pressed charges from equations (5)–(7), which also used 0.05C2F4 as the activating additive, did not combust. Meanwhile, the charges from equations (8) and (9), containing 0.1C2F4 and 0.2C2F4 respectively, did combust, but the resulting phase composition deviated significantly from the theoretical one. The products contained considerable amounts of free silicon and less SiC than expected. The recorded maximum gas pressures in the reactor – Pmax = 3.00 and 3.29 MPa – were either equal to or only slightly higher than the initial pressure P0 = 3.00 MPa. The relatively small mass losses during combustion (Δm = 15.0 and 9.9 %) also point to the low combustion intensity of charges using carbiding mixture (A). (The observed match between the maximum gas pressure and the initial pressure – Pmax = P0 = 3.00 MPa – can be by the concurrent increase in nitrogen gas pressure in the reactor due to the combustion-induced temperature rise and the simultaneous decrease in the amount of gaseous nitrogen caused by its significant uptake during the formation of silicon nitride).
It is worth noting that in a similar case of synthesizing a different composition – AlN–SiC – via azide-assisted SHS with the use of PTFE, all bulk and pressed charges based on carbiding mixture (A) underwent combustion, and did so intensively, reaching maximum reactor pressures ranging from 3.51 to 4.38 MPa. These conditions yielded good results in the formation of the target phases AlN and SiC [31]. This outcome is explained by the fact that, in the charges intended for AlN–SiC synthesis, the 0.05C2F4 PTFE additive was combined with only 1 mol of poorly reactive Si powder and 1 to 4 mol of highly reactive Al powder.
The Table shows that when using carbiding mixture (B), which contains an increased amount of the activating PTFE additive – 0.1C2F4 per 1 mol of carbiding carbon – in the charges for reactions (10)–(14), only the charges for reaction (10) fail to combust, both in bulk and pressed form. This is due to the presence of the highest amount (13 mol) of poorly reactive silicon powder in these mixtures. In reactions (11)–(14), the silicon content decreases to 4–7 mol, and the corresponding charges undergo combustion. The combustion of bulk mixtures is more intense, characterized by a somewhat higher pressure rise and greater mass loss. In some Si3N4 : SiC molar ratios, the phase composition of the target products from bulk charges more closely approximates the theoretical composition than that of the pressed charges, although up to 3 % of free silicon impurity is still present.
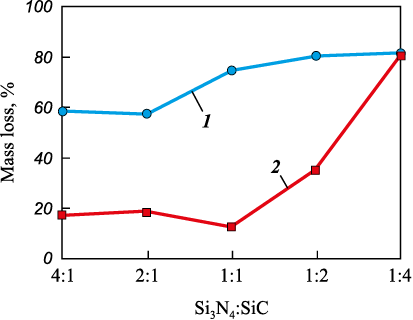
Finally, the Table shows that when using carbiding mixture (C), which contains the highest amount (0.15C2F4 ) of the activating PTFE additive per 1 mol of carbiding carbon for reactions (15)–(19), all the corresponding charges undergo intense combustion in both bulk and pressed form, reaching similar maximum reactor pressures ranging from 3.50 to 4.36 MPa. The mass loss strongly depends on the Si3N4 : SiC molar ratio. At a 1:4 ratio, the mass loss reaches very high values (up to 80 %) for both bulk and pressed charges. For other Si3N4 : SiC ratios, the mass loss remains high for bulk charges (from 57.0 to 80.1 %) but is relatively low for pressed charges (from 12.6 to 35.3 %). The composition of the washed combustion products obtained from the pressed charges is significantly closer to the theoretical values compared to that of the bulk charges, both in terms of Si3N4 and SiC phase content at all ratios, and in terms of lower levels of free silicon impurity: up to 2.0 % for pressed charges versus up to 4.0 % for bulk charges. The mass loss during combustion and the SiC phase content in the washed products obtained using carbiding mixture (C) are presented graphically in Figs. 1 and 2.
Fig. 1. Mass loss during combustion of bulk (1) and pressed (2) charges
Fig. 2. Relative theoretical (1) and experimental (2, 3) contents |
As shown in Fig. 1, the mass loss during combustion of the pressed mixtures is significantly lower than that of the bulk mixtures and becomes nearly identical – and very high – only at the Si3N4 : SiC molar ratio of 1:4, reaching 80.4 and 81.4 %, respectively. However, an experiment involving the combustion of this pressed mixture at a higher initial nitrogen pressure in the reactor (increased from 3 to 4 MPa) demonstrated a substantial reduction in mass loss – by a factor of two – to 41.9 %, while maintaining a similar phase composition in the washed combustion product: α-Si3N4 = 42.0 wt. %, β-Si3N4 = 51.0 wt. %, SiC = 6.0 wt. %, Si = 1.0 wt. %.
As evident from the Table and Fig. 2, the phase composition of the washed combustion products of the pressed charges containing PTFE in carbiding mixture (C) shows the best agreement with the theoretical phase composition in terms of Si3N4 and SiC content across all investigated Si3N4 : SiC molar ratios. This experimentally obtained phase composition is much closer to the theoretical composition of Si3N4–SiC composites than the results previously reported for conventional azide SHS (without PTFE), which yielded a significantly lower SiC content in the combustion products compared to the theoretical values [23–25].
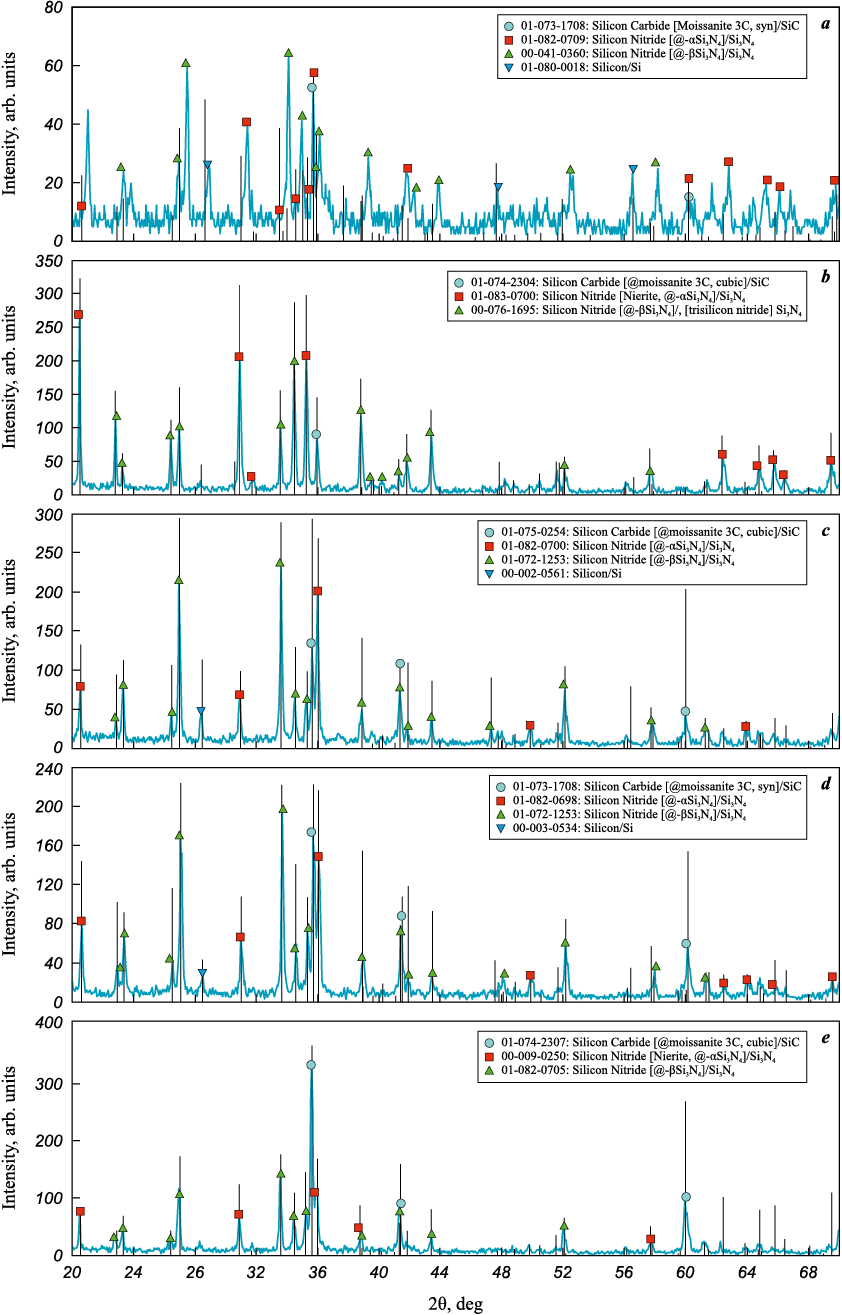
The X-ray diffraction (XRD) patterns obtained from the washed combustion products of the pressed charges synthesized using carbiding mixture (C) are shown in Fig. 3.
Fig. 3. XRD patterns of combustion products from pressed charges (15)–(19) |
The XRD patterns display strong reflections from the target phases Si3N4 and SiC, as well as either weak reflections from residual free Si or none at all. Silicon nitride is present in two polymorphic forms: α-Si3N4 and β-Si3N4 . As shown in Fig. 3 and the table, the contents of these phases are approximately equal at Si3N4 : SiC molar ratios of 4:1 and 1:4. At other ratios, however, the α-phase content is roughly half that of the β-phase. Overall, the α-phase constitutes a significant fraction – ranging from 30 to 50 % – of the silicon nitride phase in the synthesized Si3N4–SiC composites.
The microstructure of the synthesized composites is presented in Fig. 4.
Fig. 4. Microstructure of the combustion products from pressed charges (15)–(19) |
As shown in Fig. 4, the combustion products of all pressed charges are predominantly composed of highly dispersed particles smaller than 1–2 μm. The combustion product of the charge from reaction (15) with a Si3N4 : SiC molar ratio of 4:1 contains a significant fraction of columnar crystals with a transverse size of approximately 500 nm and a length of up to 2 μm, along with a small amount of finer equiaxed particles and nanofibers with diameters below 100 nm. The combustion product of the charge from reaction (16) with a 2:1 ratio includes a large fraction of coarser columnar crystals, approximately 1 μm in diameter and up to 5 μm in length, as well as equiaxed particles up to 2 μm and a small fraction of nanofibers. The product from reaction (17) with a 1:1 ratio stands out for its large proportion of nanofibers in a mixture with equiaxed particles up to 1 μm in size. The combustion product of reaction (18) with a 1:2 ratio primarily consists of agglomerates of relatively fine equiaxed particles ranging from 100 nm to 0.5 μm. Finally, the combustion product of reaction (19) with a 1:4 ratio mainly contains agglomerates of larger equiaxed particles, ranging from 200 nm to 1 μm.
Conclusion
The conventional approach of azide-assisted SHS using halide salts such as NH4F, Na2SiF6 , and (NH4)2SiF6 as activating gasifying additives previously enabled the synthesis of highly dispersed (˂1 μm) Si3N4–SiC powder compositions through combustion in a nitrogen atmosphere from mixtures of silicon powder, technical carbon, sodium azide, and a halide salt. These compositions exhibited a high content of the α-phase of silicon nitride. However, their phase compositions significantly deviated from the theoretical values, showing a much higher content of silicon nitride and a markedly lower content of silicon carbide, along with notable impurities of free silicon (up to 5.7 wt. %) and free carbon (up to 5.1 wt. %) [23–25].
In the present study, polytetrafluoroethylene (PTFE) was used instead of halide salts as both an activating and carbiding additive – partially replacing technical carbon – while sodium azide served as the nitriding additive in an amount sufficient to neutralize the fluorine released during complete PTFE decomposition. This approach facilitated the combustion of silicon–carbon powder mixtures in a nitrogen atmosphere and significantly increased the silicon carbide content in the synthesized Si3N4–SiC product, while reducing the amounts of free silicon and carbon impurities. The most favorable results were obtained using carbiding mixture (C), which contained the highest PTFE amount (0.15C2F4 + 0.7C). This can be attributed to the higher content of poorly reactive silicon powder (4 to 13 mol) in the charges for synthesizing Si3N4–SiC compositions compared to, for example, a similar PTFE-based azide SHS process for AlN–SiC compositions. In the latter case, the best results were achieved using carbiding mixture (A) with the lowest PTFE content (0.05C2F4 + 0.9C) in charges containing only 1 mol of poorly reactive Si powder and 1 to 4 mol of highly reactive Al powder.
Based on the obtained data on mass loss during combustion and on the phase composition of the combustion products, it can be recommended that Si3N4–SiC powder compositions be synthesized via combustion of pressed charges in azide SHS with partial replacement of 0.3 mol of carbon by 0.15 mol of PTFE, at an initial nitrogen pressure of 3 MPa (or 4 MPa to reduce mass loss when the Si3N4 : SiC phase ratio is 1:4). The resulting combustion products are predominantly mixtures of highly dispersed particles smaller than 1–2 μm and contain a substantial fraction (30–50 %) of the α-phase in the silicon nitride component of the synthesized Si3N4–SiC compositions. The phase composition of these products is significantly closer to the calculated theoretical composition of the target Si3N4–SiC system than that achieved using conventional azide SHS without PTFE.
References
1. Li J., Yuan W.J., Deng C.J., Zhu H. X. The effect of Si contents on the reaction-bonded Si3N4/SiC composite ceramics. IOP Conference Series: Materials Science and Engineering. 2013;47(1):012060. https://doi.org/10.1088/1757-899X/47/1/012060
2. Chenhong M., Li Y., Jiang P., Yue X. Synthesis mechanism of α-Si3N4 whiskers via SiO vapor in reaction bonded Si3N4–SiC composite. Journal of Alloys and Compounds. 2023;938(6):168723. https://doi.org/10.1016/j.jallcom.2023.168723
3. Karklit A.K., Porinsh N.M., Katorgin G.M., Norkina A.S., Solominskaya I.Yu. Refractory products, materials and raw materials. Moscow: Metallurgiya, 1990. 416 p. (In Russ.).
4. Strelov K.K. Theoretical foundations of refractory materials technology. Moscow: Metallurgiya, 1985. 480 p. (In Russ.).
5. Surendranathan A.O. An introduction to ceramics and refractories. 1st ed. Boca Raton: CRC Press, Taylor & Francis Group, 2015. 524 p. https://doi.org/10.1201/b17811
6. Shahrestani S., Ismail M., Kakooei S., Beheshti M. Microstructure, phase compositions and mechanical properties of slip cast sintered SiC/Si3N4 composites. Ceramics International. 2021;47(12):13173–13180. https://doi.org/10.1016/j.ceramint.2021.01.182
7. Yang H., Li Q., Wang Zh., Sun M., Wu H., Cheng X. Effect of different sintering additives on the microstructure, phase compositions and mechanical properties of Si3N4/SiC ceramics. ES Materials and Manufacturing. 2022;15:65. https://doi.org/10.30919/esmm5f487
8. Saleem A., Zhang Yu., Gong H., Majeed M.K. Fluoride doped SiC/Si3N4 composite as a high thermal conductive material with enhanced mechanical properties. Ceramics International. 2019;45(16):21004–21010. https://doi.org/10.1016/j.ceramint.2019.06.289
9. Khajelakzay M., Bakhshi S.R. Optimization of spark plasma sintering parameters of Si3N4–SiC composite using response surface methodology (RSM). Ceramics International. 2017;43(9):6815–6821. https://doi.org/10.1016/j.ceramint.2017.02.099
10. Wang J.-Ch., Dommati H., Hsieh Sh.-J. Review of additive manufacturing methods for high-performance ceramic materials. The International Journal of Advanced Manufacturing Technology, 2019;103(1–4):2627–2647. https://doi.org/10.1007/s00170-019-03669-3
11. Basu B., Balani K. Advanced structural ceramics. Hoboken, New Jersey: John Wiley & Sons, Inc., 2011. 502 p.
12. Niihara K. New design concept of structural ceramics-ceramic nanocomposites. Journal of the Ceramic Society of Japan. 1991;99(1154):982974–982982. https://doi.org/10.2109/jcersj.99.974
13. Hirano T., Niihara K. Microstructure and mechanical properties of Si3N4/SiC composites. Materials Letters. 1995;22(5–6):249–254. https://doi.org/10.1016/0167-577X(94)00255-X
14. Palmero P. Structural ceramic nanocomposites: A review of properties and powders’ synthesis methods. Nanomaterials. 2015;5(2):656–696. https://doi.org/10.3390/nano5020656
15. Montanaro L., Palmero P. Advances in the field of nanostructured ceramic composites. Ceramics. 2019;2: 296–297. https://doi.org/10.3390/ceramics2020024
16. Li X., Lei W., Wei J., Zhang Ya., Shi Z. Synthesis and broadband electromagnetic wave absorption for lightweight porous SiC/Si3N4 composite nanowires. Ceramics International. 2023;49(23):37746–37757. https://doi.org/10.1016/j.ceramint.2023.09.102
17. Riedel R., Seher M., Becker G. Sintering of amorphous polymer-derived Si, N and C containing composite powders. Journal of The European Ceramic Society. 1989;5(2):113–122. https://doi.org/10.1016/0955-2219(89)90018-6
18. Yanai T., Ishizaki K. Mechanical properties of Si3N4 ceramics prepared from carbon coated powders. Journal of the Ceramic Society of Japan. 1993;101(1175):764–768. https://doi.org/10.2109/jcersj.101.764
19. Suri J., Shaw L., Zawrah M.F. Tailoring the relative contents of Si3N4 and SiC through carbothermic reduction and nitridation of silica fume. International Journal of Applied Ceramic Technology. 2012;9(2):291–303. https://doi.org/10.1111/j.1744-7402.2011.00710.x
20. Hojo J., Meada H., Kato A. Preparation of composite particles of SiC–Si3N4 system by vapor reaction method. Yogyo-Kyokai-Shi. 1987;95(1):45–49.
21. Lee H.J., Eguchi K., Yoshida T.J. Preparation of ultrafine silicon nitride, and silicon nitride and silicon carbide mixed powders in a hybrid plasma. Journal of the American Ceramic Society. 2005;73(11):3356–3362. https://doi.org/10.1111/j.1151-2916.1990.tb06461.x
22. Khachatryan G.L., Arutyunyan A.B., Kharatyan S.L. Activated combustion of a silicon–carbon mixture in nitrogen and SHS of Si3N4–SiC composite ceramic powders and silicon carbide. Combustion, Explosion, and Shock Waves. 2006;42(5):543–548. https://doi.org/10.1007/S10573-006-0086-7
23. Amosov A.P., Belova G.S., Titova Yu.V., Maidan D.A. Synthesis of highly dispersed powder ceramic composition Si3N4–SiC by combustion of components in the Si–C–NaN3–NH4F system. Russian Journal of Inorganic Chemistry. 2022;67(2):123–130. https://doi.org/10.1134/S0036023622020024
24. Amosov A.P., Titova Yu.V., Belova G.S., Maidan D.A., Minekhanova A.F. SHS of highly dispersed powder compositions of nitrides with silicon carbide. Review. Powder Metallurgy and Functional Coatings. 2022;16(4):34–57. https://doi.org/10.17073/1997-308X-2022-4-34-57
25. Belova G.S. Self-propagating high-temperature synthesis of ceramic nitride-carbide highly dispersed powder compositions Si3N4–SiC, AlN–SiC and TiN–SiC using sodium azide and halide salts: Diss. Cand. Sci. (Eng.). Samara: SamSTU, 2022. 209 p. (In Russ.).
26. Mukasyan A.S. Combustion synthesis of silicon carbide. In: Properties and applications of silicon carbide. Ed. R. Gerhardt. Rijeka, Croatia: InTech, 2011. P. 361–388.
27. Amirkhanyan N., Kirakosyan H., Zakaryan M., Zurnachyan A., Rodriguez M.A., Abovyan L., Aydinyan S. Sintering of silicon carbide obtained by combustion synthesis. Ceramics International. 2023;49(15):26129–26134. https://doi.org/10.1016/j.ceramint.2023.04.233
28. Vorotilo S., Levashov E.A., Potanin A.Yu., Loginov P.A., Shvyndina N.V. Features of synthesizing ceramic composites discretely reinforced by carbon fibers and SiC nanowires formed in situ in the combustion wave. Russian Journal of Non-Ferrous Metals. 2020;61(5):559–570. https://doi.org/10.3103/S1067821220050168
29. Zakorzhevsky V.V., Loryan V.E., Akopdzhanyan T.G. Self-propagating high-temperature synthesis of silicon carbide nanofibers. Russian Journal of Non-Ferrous Metals. 2020;61(6):675–679. https://doi.org/10.3103/S106782122006022X
30. Nersisyan G.A., Nikogosov V.N., Kharatyan S.L., Merzhanov A.G. Chemical transformation mechanism and combustion regimes in the system silicon-carbon-fluoroplastic. Combustion, Explosion, and Shock Waves. 1991;27(6):720–724. https://doi.org/10.1007/BF00814517
31. Amosov A.P., Titova Yu.V., Uvarova I.A., Belova G.S. Azide self-propagating high-temperature synthesis of a highly dispersed AlN–SiC powder composition using polytetrafluoroethylene. Powder Metallurgy аnd Functional Coatings. 2024; 18(6):28–43. https://doi.org/10.17073/1997-308X-2024-6-28-43
32. Titova Yu.V., Uvarova I.A., Amosov A.P., Belova G.S., Maidan D.A. Preparation of Si3N4–SiC powder composition using SHS azide technology using polytetrafluoroethylene. In: Proceedings of 9th International Congress on Energy Fluxes and Radiation Effects (EFRE-2024) (Tomsk, Russia, 16–21 September 2024). Tomsk: TPU Publishing House, 2024. P. 1489–1494. (In Russ.). https://doi.org/10.56761/EFRE2024.N2-O-031103
About the Authors
I. A. UvarovaRussian Federation
Irina A. Uvarova – Engineer of the Department of metallurgy, powder metallurgy, nanomaterials (MPMN)
244 Molodogvardeiskaya Str., Samara 443100, Russia
A. P. Amosov
Russian Federation
Aleksandr P. Amosov – Dr. Sci. (Phys.-Math.), Prof., Head of the Department of MPMN
244 Molodogvardeiskaya Str., Samara 443100, Russia
Yu. V. Titova
Russian Federation
Yuliya V. Titova – Cand. Sci. (Eng.), Associate Prof. of the Department of MPMN
244 Molodogvardeiskaya Str., Samara 443100, Russia
V. A. Novikov
Russian Federation
Vladislav A. Novikov – Cand. Sci. (Eng.), Associate Prof. of the Department of MPMN
244 Molodogvardeiskaya Str., Samara 443100, Russia
Review
For citations:
Uvarova I.A., Amosov A.P., Titova Yu.V., Novikov V.A. Self-propagating high-temperature synthesis of a highly dispersed Si3N4–SiC ceramic powders composition using sodium azide and polytetrafluoroethylene. Powder Metallurgy аnd Functional Coatings (Izvestiya Vuzov. Poroshkovaya Metallurgiya i Funktsional'nye Pokrytiya). 2025;19(3):25-38. https://doi.org/10.17073/1997-308X-2025-3-25-38